What if the Y chromosome hadn't evolved as expected?
This is one of the best-known facts in genetics: women are XX and men are XY. In humans, chromosomes determine sex, and this particular pair, the23rd, is the most remarkable. It is the only pair of chromosomes where two versions with enormous differences coexist. So much so that it is even difficult to recognize in this pair two chromosomes that were once very closely related but which, over millions of years of evolution, have become unrecognizable.
Thomas Lenormand, University of Montpellier and Denis Roze, Sorbonne University

Why have sex chromosomes evolved to become so unique? This question has puzzled geneticists for nearly a century. Several hypotheses have been proposed over time, and one theory has gradually gained traction. However, this long-standing theoretical framework has just been upended by a new model that challenges its foundations.
The X-Y dichotomy
NIH/Flickr, CC BY-NC
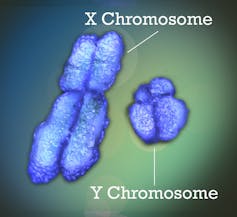
The X chromosome is fairly ordinary, containing nearly 800 protein-coding genes. The Y chromosome, on the other hand, is much more unusual: it is smaller (about one-third the length of the X) and contains only about sixty genes. Nevertheless, these two chromosomes do form a “pair.” During sperm production, they pair up and undergo recombination over a small section (recombination is an exchange of genetic material, in this case between the two chromosomes of the same pair), reflecting their ancestral homology. But 95% of the Y chromosome no longer undergoes recombination at all. The X chromosome, on the other hand, undergoes recombination along its entire length in XX females during egg production.
Finally, another peculiarity is that only one X chromosome is expressed in females. That is to say, genes are transcribed to produce proteins from a single X chromosome—either the one inherited from the father or the one inherited from the mother—randomly depending on the cell. This allows for “dose compensation,” ensuring that the genes carried on the X chromosome are expressed at the same level in both males and females.
Widespread differences in living organisms
One might think that these quirks are unique to humans. That is not the case: they are in fact extremely common among animals—and even plants—although the specifics vary.
However, we consistently observe a halt in recombination on the sex chromosomes, a non-recombining chromosome (Y) that contains few functional genes (it is “degenerate”), as well as dosage compensation.
These patterns quickly caught the attention of geneticists, who began searching for a theory to explain why recombination stops on these chromosomes, and why this halt in recombination is associated with degeneration. These same geneticists also quickly realized that answering this question might also shed light on a broader issue that puzzled them even more: the role of recombination in evolution.
The role of recombination
Recombination, which occurs during gamete formation, facilitates genetic “shuffling.” It is an essential feature of sexual reproduction and is undoubtedly the key factor explaining the advantage of sex over clonal asexual reproduction. The degeneration of non-recombining sex chromosomes appears to be a real-world demonstration of the importance of recombination in maintaining genomic integrity in the face of a constant stream of deleterious mutations that alter genetic information.
In fact, most chromosomes carry mutations, which can be passed down from one generation to the next when their effect on the survival or fertility of organisms is not too significant. The mutation-free version of a chromosome segment may be quite rare within a population, and may even disappear if the individuals carrying it do not produce offspring. When this happens, a mutation-free segment can only be recreated through recombination between chromosomes carrying mutations at different locations. Because of this process, we expect that the non-recombining regions of chromosomes will gradually accumulate mutations.
The lack of recombination could therefore explain the degeneration of the Y chromosome. But that doesn’t fully answer the question: why on earth do sex chromosomes stop recombining if doing so leads to degeneration?
The theory of "sex-antagonistic" genes
Until our research, this was explained by the theory of “sex-antagonistic” genes. Males and females differ in many traits: this is known as sexual dimorphism. For many genes, there are therefore versions (called “alleles”) that are advantageous for one sex and disadvantageous for the other.

benjamint444/Wikimedia, CC BY-SA
If these "sex-antagonistic" genes are common, we would expect to find them on the sex chromosomes. Suppressing recombination (and thus the exchange of alleles between the X and Y chromosomes) can then become advantageous. If an allele that is advantageous in males is found on a Y chromosome that no longer recombines, it will remain on that Y chromosome: it will always be present in males but never in females.
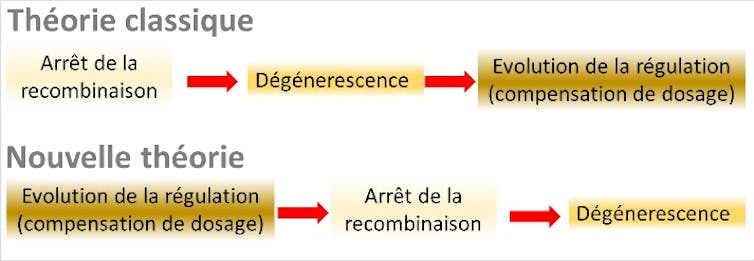
A three-stage global theory of sex chromosome evolution has thus emerged. Stage 1 is the cessation of recombination on the Y chromosome, caused by the presence of sex-antagonistic genes on the sex chromosomes. Stage 2 is the accumulation of deleterious mutations (degeneration), resulting from the cessation of recombination. Stage 3 is the evolution of dosage compensation to compensate for the deficit in X-gene expression in males.
This theory had the advantage of being very elegant, as it was highly general. However, several subsequent observations gradually called this framework into question and reignited the theoretical debate.
A new theory
To this end, we started with the only "ingredients" that appear essential for explaining the evolution of sex chromosomes: events that halt recombination on the Y chromosome (though these are not irreversible), deleterious mutations (degeneration cannot occur without them), and gene expression regulators that can modulate gene expression levels (dose compensation cannot occur without them).
To our great surprise, these ingredients—and particularly the last one, which had never been formally incorporated into classical theory—proved to be sufficient. Our research has revealed an overarching process that is entirely different from “classical” theory, in which the entire set of causal relationships is reversed, and “sex-antagonistic” genes become redundant.
Thomas Lenormand, Courtesy of the author
Like classical theory, this new theory posits a primordial scenario in which a gene carried by a pair of “normal” chromosomes (autosomes) determines an individual’s sex. Two different forms of this gene (or alleles, denoted M and F) coexist, with MF individuals developing into males and FF individuals into females.
We then assume that chromosomal inversions can occur: an inversion involves a “flipping” of a portion of the chromosome, with the genes located in that portion ending up in reverse order. These inversions represent a relatively rare form of mutation, but they do occur from time to time within populations. They prevent (at the site of the inversion) recombination between inverted and non-inverted chromosomes. Indeed, the different order of the genes prevents the chromosomes from pairing correctly in this region.
When an inversion involving the M allele (which determines male sex) occurs, it may be favored if the genes it contains carry fewer deleterious mutations than average. In this case, its frequency will increase over generations until all male individuals carry this inversion. As a result, the chromosomes carrying the F and M alleles become “proto-chromosomes” X and Y, no longer recombining over a portion of their length (in the inversion zone).
However, the cessation of recombination eventually leads to an accumulation of deleterious mutations on the chromosome carrying the M allele (since the chromosome carrying the F allele continues to recombine in females). This accumulation of mutations can either promote a return to recombination (for example, through a new chromosomal inversion restoring the initial order of the genes) or a reduction in the expression of the mutated genes carried by the proto-Y, which will be compensated for by an increase in the expression of the proto-X genes: this marks the beginning of dosage compensation. When this process establishes itself quickly enough, it prevents any reversal: a restoration of recombination would destroy this delicate balance between the expression levels of the X and Y genes.
The computer simulations we conducted thus show a gradual cessation of recombination, occurring in successive “layers” corresponding to different inversions stabilized by changes in dosage compensation, without the need to invoke the existence of genes with sex-antagonistic effects.
By completely transforming our understanding of possible evolutionary scenarios for sex chromosomes, this new theory opens up a vast field of experimentation and empirical testing. It also highlights the importance of incorporating mechanisms that regulate gene expression into evolutionary theory. Finally, it suggests that considering the evolution of regulators could be one of the keys to solving the mystery of why sexual reproduction persists in plants and animals.![]()
Thomas Lenormand, Research Director, University of Montpellier and Denis Roze, Researcher in evolutionary genetics, Sorbonne University
This article is republished from The Conversation under a Creative Commons license. Readthe original article.