Intracellular protein interactions may soon be explained
How do proteins interact within cells? This is a complex question that may soon be answered thanks to the work of an international consortium led by Jérémie Torrès, Professor the Institute of Electronics and Systems (IES) at the University of Montpellier. A winner of the prestigious European FET-Open call for proposals, this project focuses on developing a biosensor capable of directly measuring these interactions inside living cells.
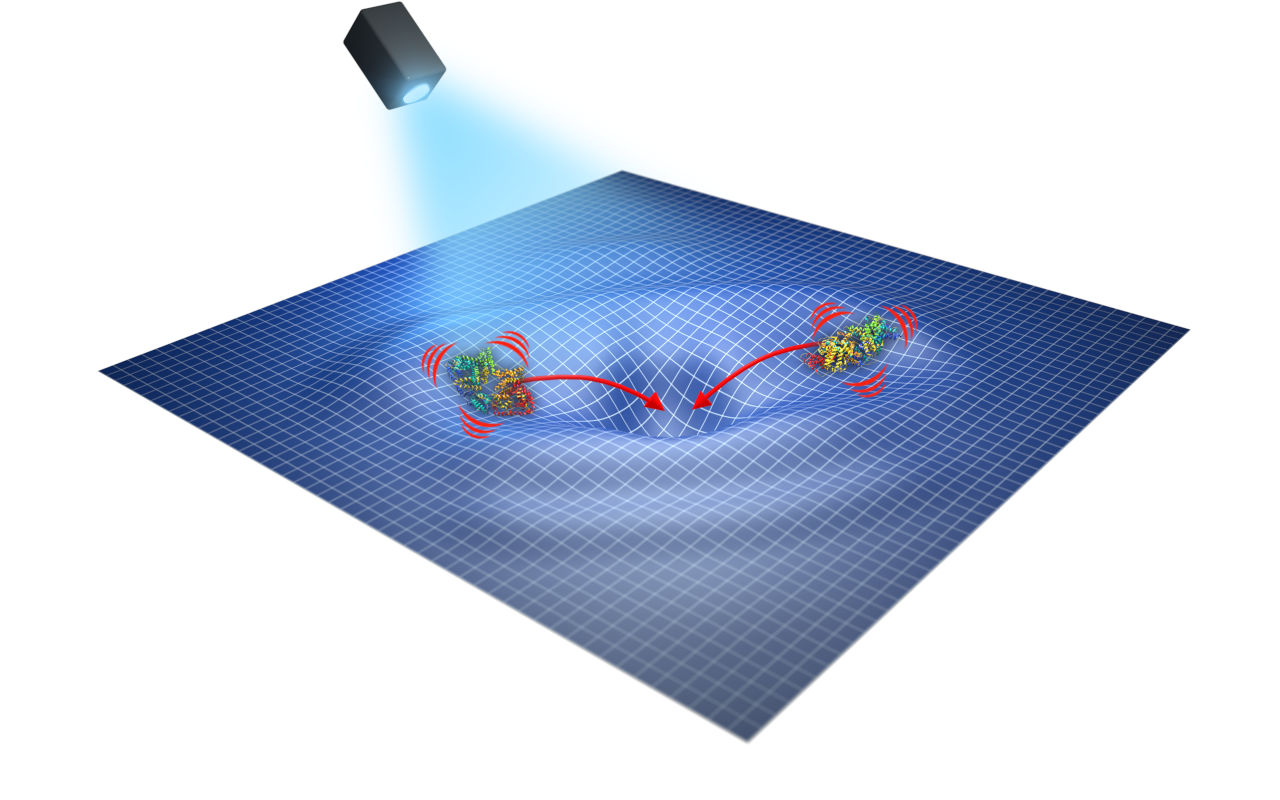
Pour un public averti, cette question ne date pas d’hier. Prédites dès le début du XXème siècle par l’électrodynamique classique et quantique, les forces intermoléculaires agissant à longue distance (<1000 angströms) n’ont jamais été démontrées expérimentalement. Plus pour longtemps, comme l’explique Jérémie Torrès, enseignant-chercheur à l’Institut d’Électronique et des Systèmes (IES) : « Nous avons trouvé un système d’étude qui permet de démontrer ces interactions entre molécules, qui se trouvent être des protéines. »
The“we”used by Jérémie Torres refers to a consortium comprising four French laboratories under the auspices of the CNRS, including two in Montpellier [the IES and the Charles Coulomb Laboratory (L2C)], one German laboratory, one Swedish laboratory, and two companies: the Montpellier-based startup TeraKalis and the Netherlands-based company Micronit. Under the leadership of the Montpellier-based researcher and his colleague at L2C, Sandra Ruffenach, the project was submitted to the prestigious European FET-Open call for research and innovation actions (RIA)… and selected, receiving the highest possible score!
Coincidence or design?
It all starts with an observation: when proteins move through the cytoplasm, they collide with one another and are involved in a vast number of biomolecular reactions over a very short period of time. “It’s a bit like when you start a game of pool with the break shot—the balls fly off in all directions at random,” explains Jérémie Torres. “This is what’s known as Brownian motion.” While this observation has long been accepted as dogma, it poses a problem: how can these countless reactions be effective if they are guided solely by chance?
This is the problem that Jérémie Torres and his colleagues set out to tackle. “We’re not the first. Back in the 1960s, the English physicist Herbert Fröhlich had already raised it.” Biology has, in fact, already demonstrated how, over short distance scales (less than ten angstroms), forces well known in physics govern these intermolecular interactions. “The Coulomb forces Coulomb, vyearr Waals, etc., explain how proteins, when they are very close to one another, end up attracting each other and sticking together to trigger reactions. But what we don’t know is why they came together in the first place. Are there mechanisms that allow them to‘callout’ to one another?” asks the IES researcher.
Spectral signature
Thanks to the 3.1 million euros in funding provided by the European Union through the FET-Open program, researchers will be able to work on developing a new technology to measure these interactions.“We are working on a biosensor prototype that will allow us to measure these interactions based on the terahertz spectral signature of proteins in an aqueous environment. We are currently the only ones in the world with this expertise.”
Since each protein has its own unique spectral signature, this could enable its non-invasive identification. To measure it, the researchers will, in a sense, force the proteins to synchronize their oscillations,“like metronomes that are forced to oscillate at the same time,” adds Jérémie Torrès.“When proteins oscillate collectively, they resonate at a specific frequency within the terahertz range. We will measure these resonances to highlight the long-range intermolecular interactions that could underlie the mechanisms governing molecular dynamics within cells.”
Physical Sciences vs. Science Fiction
While this research project is primarily focused on fundamental and technological aspects—namely, gaining a better understanding of the mechanisms of protein-protein interactions—it could also, in the long term, lead to major therapeutic innovations. How? By influencing these mechanisms, or even inhibiting them, when they are involved in the development of certain diseases.“The dream would indeed be to establish the role played by a protein in a disease and to know the frequency at which it resonates; we could then imagine destroying this protein with an electromagnetic wave and thus blocking the entire disease development process, all while being non-destructive to the rest of the body. But that’s still science fiction,”concludes Jérémy Torres.