Mnemonics: When Memory Involves Prion Proteins
Just as we are able to store information in our memory, single-celled organisms can retain memories of past stressors. This allows these simple cells to respond more effectively to the same stressors in the future—and thus, for example, ensure the colony’s survival.
Fabrice Caudron, University of Montpellier
But how are these memories formed at the molecular level? Can these mechanisms teach us more about the very foundations of memory, even in humans? Or even help us better understand the mechanisms behind certain diseases?
To learn more, we investigated a memory process found in baker’s yeast, Saccharomyces cerevisiae. Here are the results of our research, which have just been published in the journal Current Biology.
Sex and memory in baker's yeast
Saccharomyces cerevisiae is a fairly simple organism: consisting of a single cell with a nucleus (like human cells and unlike bacteria), it reproduces asexually, meaning that the cell forms a bud on its surface that will develop into a daughter cell.
However, S. cerevisiae can also reproduce sexually. To identify a potential sexual partner, yeast cells secrete and detect specific chemical substances: sex pheromones. When two yeast cells of opposite sexes come into close proximity, they cease vegetative reproduction and extend a cytoplasmic projection (the cytoplasm being the “body” of the cell, the part surrounding the nucleus), known as a “shmoo,” toward their counterpart.
Since yeast cells cannot move, they extend toward one another until they meet and fuse into a new cell, where their genomes mix. This increases their genetic diversity.
But this commitment to sexual reproduction is a costly, if not risky, decision. For example, imagine that two cells of the same sex are courting a single cell of the opposite sex. One of these two cells will be unable to fuse with its prospective partner and would therefore have produced a shmoo for nothing… Fortunately, yeast cells can “choose” not to respond to the pheromone—a refusal they will then remember for a long time.
In fact, once a yeast cell stops responding to the pheromone, it remains that way for the rest of its life. From then on, it will reproduce only asexually. In contrast, its future daughter cells will be “naive”: they do not inherit their mother’s memory and are capable of responding to any sex pheromone present in their environment.
How does the memory of a single-celled organism work?
The key to S. cerevisiae’s memory is the Whi3 protein. By changing its conformation (the way it folds in three dimensions), Whi3 becomes inactive, allowing the cell to ignore the pheromone.
We have observed that the Whi3 protein behaves somewhat like a prion —a pathogenic protein due to its abnormal 3D folding, and one that is often contagious. Prion-like proteins were first identified in the 1980s by the American neurologist Stanley Prusiner, particularly in the wake of the mad cow disease crisis: he demonstrated that by changing its three-dimensional conformation, the prion protein becomes pathogenic and contagious, causing not only the disease in cattle but also Creutzfeldt-Jakob disease in humans.
Returning to yeast, the conformational change in Whi3 is self-replicating; that is, it spreads from an abnormal protein to a normal protein (which it “contaminates”) through simple contact, forming aggregates. This self-replicating phenomenon for encoding memory is interesting because it implies that the new conformation of the Whi3 protein can be established over time through transmission and remain stable.
However, a major problem remains: how can the parent cell ensure that “its memory” does not spill over into its daughter cell?
Understanding this type of physical memory
It is important to note that yeast contains many types of prion proteins, most of which, once formed, are transmitted to the daughter cell. This is not the case with Whi3, however, which is what makes it unique.
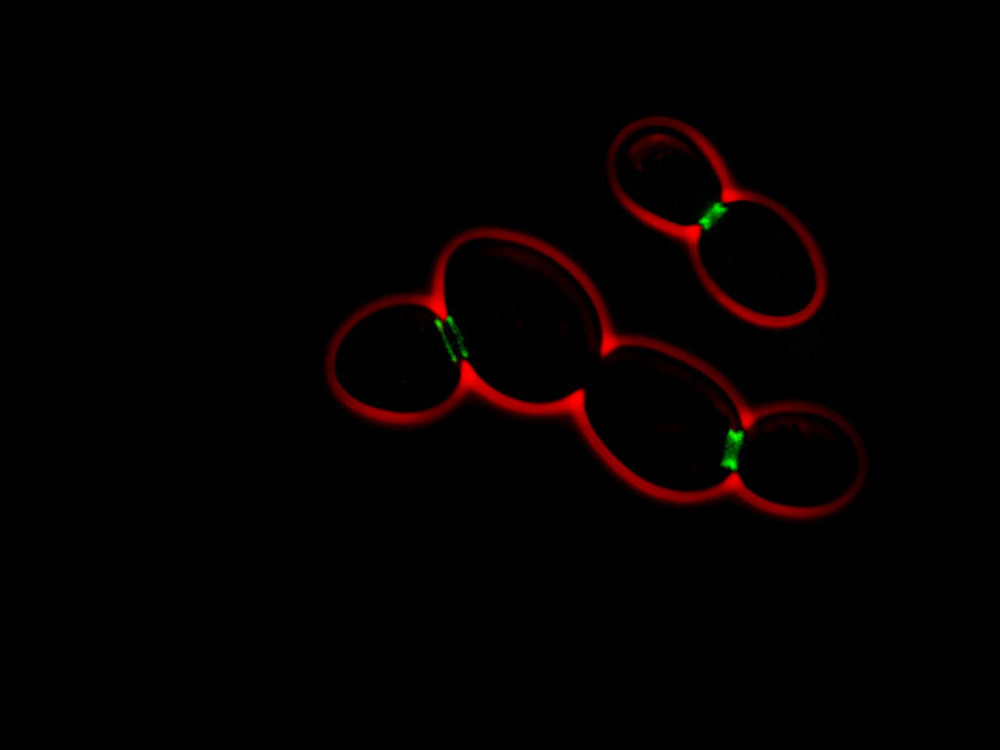
Using yeast genetics and microscopy, we compared the best-understood yeast prion, called Sup35 (the translation termination factor), with Whi3.
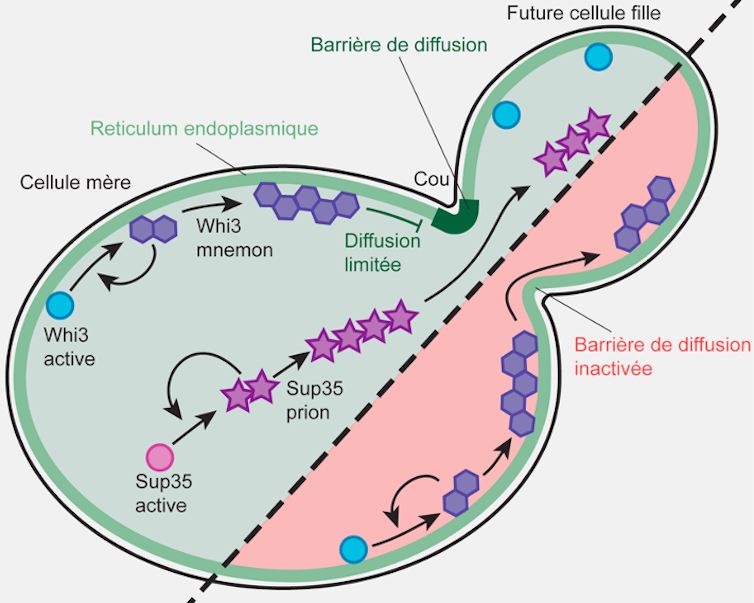
We have already discovered that Whi3 binds to the membranes of the endoplasmic reticulum (a vast network of sacs where the cell assembles its proteins). We have also found that, within this reticulum, barriers are established between the mother cell and the components of its future daughter cell—thus allowing the mother cell’s Whi3 to be retained.
Genetic removal of diffusion barriers causes Whi3 to transform into a “true” prion, which can spread to daughter cells. The confinement of Whi3 and the memory encoded by this protein are lost. In contrast, the spread of the Sup35 prion, which does not bind specifically to the endoplasmic reticulum, is not affected by the diffusion barrier.
Fabrice Caudron, Provided by the author
A step toward a better understanding of cellular memory
This study highlights that the association of prion-like proteins with membranes, together with the compartmentalization of these membranes by diffusion barriers, constitutes a powerful mechanism for the formation of long-term epigenetic cellular memories (i.e., memories not linked to the information encoded in DNA). These “memories” can be confined to a subcompartment of the cell, in this case the dividing mother cell.
The Whi3 protein, although very similar to prions, is therefore considered a “mnemon”: a specific type of prion that is kept under control and encodes a memory. But, ultimately… what does this have to do with neural memory—the kind at work in our brains?
It turns out that a protein important for neural memory—for example, during the encoding of sexual experiences in the fruit fly— depends on the prion-like behavior of the CPEB protein in the synapse, the connection point between two neurons.
We hypothesize that in Drosophila, CPEB is also a mnemonic protein, confined to the synapse that is activated during memory formation. This confinement would prevent the CPEB protein from diffusing into neighboring synapses of the same neuron (which could cause them to fire erratically and compromise memory formation).
In Homo sapiens, the CPEB3 protein exhibits the same characteristics as Drosophila’s CPEB: it can behave like a prion, or more likely, a mnemon. These similarities suggest that the cellular basis of memory has a long evolutionary history…
These findings and hypotheses raise the question of whether neurodegenerative diseases associated with prions and protein aggregates might sometimes be caused by defects in cellular containment. We know, for example, that these barriers are less effective in stem cells from aged mice. Might it therefore be feasible to attempt to restore them in order to limit the spread of prion-like proteins?
This is still just a theoretical idea, but it opens up many possibilities…![]()
Fabrice Caudron, Team Leader, Cell Biology, Yeast Genetics, Asymmetric Cell Division, Cellular Memory, University of Montpellier
This article is republished from The Conversation under a Creative Commons license. Readthe original article.