Tardigrades can withstand (almost) anything, thanks to genes from extinct species
Tardigrades are small but tough: X-rays, extreme temperatures, immense pressure—they demonstrate incredible resilience. A new study shows that as they evolved, they acquired genes from other species that gave them these “superpowers.”
Simon Galas, University of Montpellier and Myriam Richaud, University of Montpellier
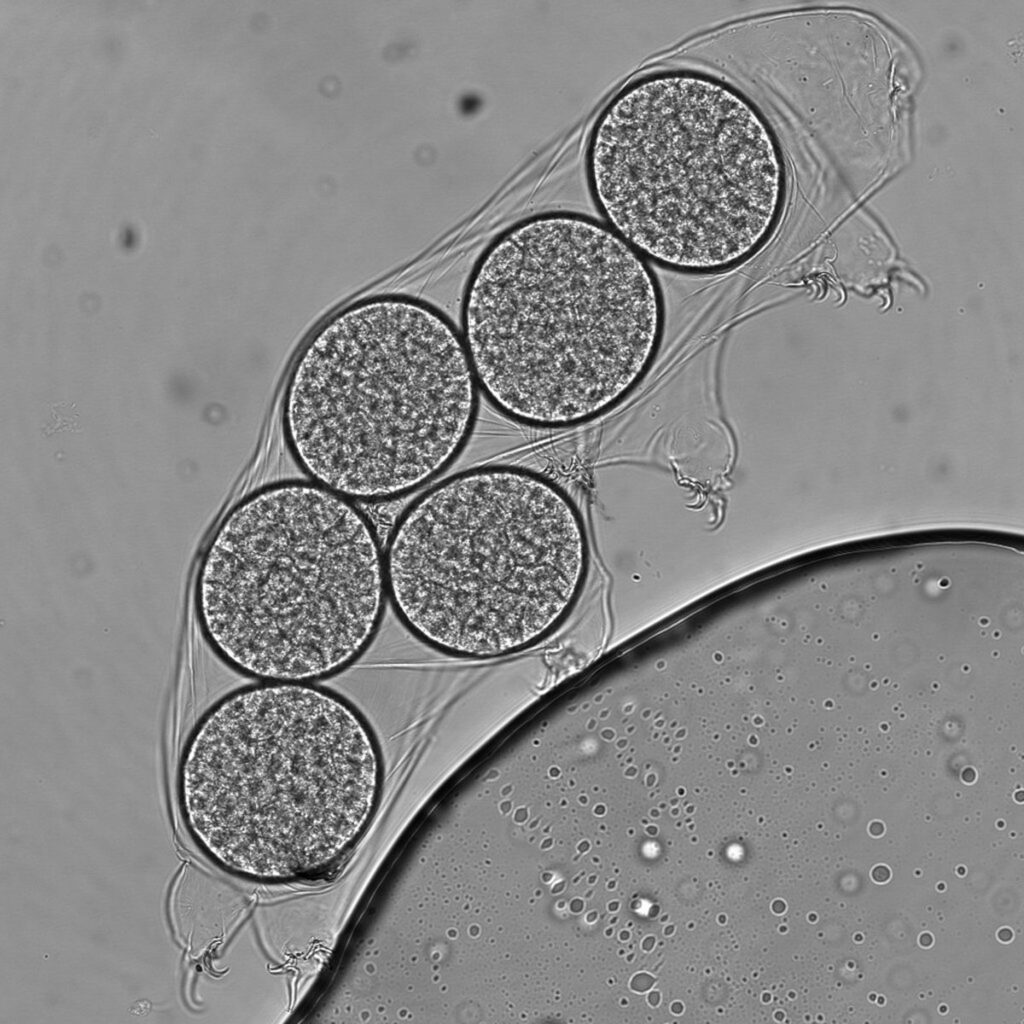
Imagine a tiny organism found everywhere on our planet, all around us, carrying with it a lost genetic memory. Tardigrades are invertebrates measuring 0.2 to 1.2 millimeters in length that resemble tiny bears with four pairs of legs, muscles, neurons, and a microbiome. They can be found everywhere on our planet, from the ocean floor to the summit of the Himalayas.
Tardigrades are also known as water bears because they always live in environments where water is present, such as oceans, glaciers, rivers, or house gutters, as well as in mosses and lichens on trees or rocks. Nearly 1,500 species are already known, and they are the survival champions of our planet and the undisputed kings of a very exclusive club known as extremophiles—organisms capable of surviving in the most extreme environments.
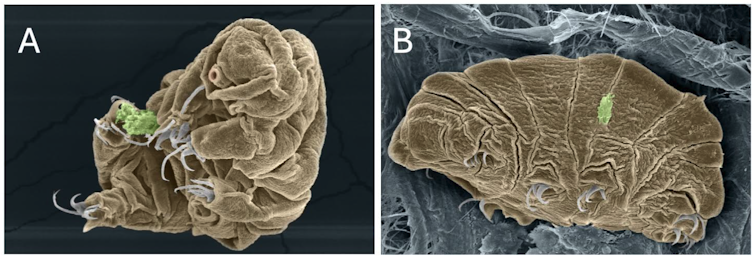
In fact, tardigrades are capable of withstanding the lowest temperature ever recorded in the universe (-272 °C) as well as temperatures close to those found on Mercury (+151 °C). They can even survive temperatures close to absolute zero (-273.16 °C), which does not exist in the universe but only in physics laboratories. In a recent physics experiment, one of the three tardigrades of the species Ramazzotius varieornatus, pictured below, was successfully revived after exposure to a temperature close to absolute zero.
Tardigrades can also survive a ten-day stay in the vacuum of space while directly exposed to cosmic rays and have become a model organism for research in astrobiology. As for radiation, we know they can survive X-ray doses 1,000 times higher than those that are lethal to humans. Their resistance to extreme pressure has also been tested for several hours, and, surprisingly, they survive being crushed by a weight equivalent to a building with… 60,000 floors.
Cryptobiosis: Life on Hold
The first discovery of tardigrades dates back tothe 18thcentury. After studying with the Jesuits in Reggio (Calabria, Italy), the biologist and philosopher Lazzaro Spallanzani (1729–1799) published the first study on these tiny creatures in 1776 in his work *Opuscules de physique animale et végétale*. He gave them the name “tardigrade” and observed their ability to dehydrate completely and then “resurrect after death” in the presence of water, describing the phenomenon of cryptobiosis for the first time.
Cryptobiosis is a “state of suspended animation” during which no signs of life are detectable. In this state, tardigrades collected in Antarctica were successfully revived after thirty years. Other data have shown that tardigrades in cryptobiosis are not only in a “state of suspended animation” but also “outside of time.” Indeed, the time spent in this state of cryptobiosis is not counted toward their normal lifespan (the average lifespan of a tardigrade species in controlled breeding conditions is approximately 60 days). In short, whether or not it enters cryptobiosis, a tardigrade’s normal active lifespan remains unchanged. Anglophones refer to this phenomenon as “Sleeping Beauty,” indicating that an organism halts its aging process as long as it remains in this state.
Our CNRS laboratory in Montpellier was the first to successfully observe what happens inside a species of tardigrade (Hypsibius exemplaris) when it enters cryptobiosis. In this state, this species shrinks by losing 38% of its volume and builds a visible barrier around each of the cells that make up its body. This structure gradually disappears as the animal is revived.
Survival strategies that vary by species
But the most surprising finding comes from a recent study by our laboratory on a species related to the first one (Ramazzottius varieornatus), also from our breeding colonies. When it enters cryptobiosis, this species shrinks by only 32%. Even more surprising, it was impossible to observe the presence of that specific cryptobiotic barrier that surrounded the cells of the previous species. These experiments indicate that different species of tardigrades are capable of withstanding stresses that are lethal to other living species, but that they do so in different ways and using mechanisms that are not all shared among them.
Starting in 2016, this set of genetic tools that enables them to withstand extreme environments began to be identified during the first sequencing of their genomes. Scientists are already interested in these tools for future revolutionary biomedical applications, such as storing drugs and vaccines in a dehydrated form or protecting cells from lethal radiation—which would be useful for future space missions.
Geneticists believe that tardigrades acquired these genes to enable them to withstand dehydration, but they also suggest that these same genetic tools allow them to survive in all kinds of deadly environments. While studying their genetic makeup, scientists were surprised to find that nearly 40% of tardigrade genes are unknown in other species currently living on our planet.
But where do these genes, known as "unique tardigrade genes," come from?
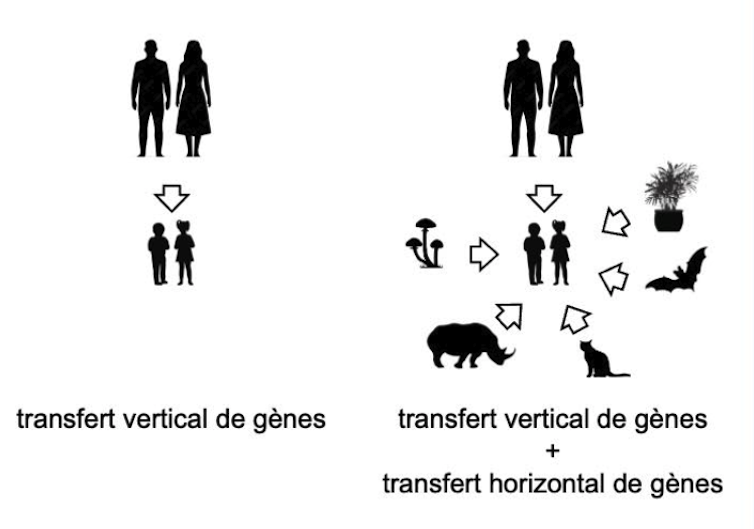
One explanation involves the mechanism of horizontal gene transfer (HGT). As illustrated below, a living organism typically inherits genes from its parents through vertical transmission.
To acquire genes from one's neighbors
In the case of horizontal gene transfer, an organism has an additional option: the ability to acquire genes from its neighbors and retain them if they prove beneficial to the survival of its species. This has already been observed in a species of aphid in which green individuals are eaten by ladybugs, while red ones are parasitized by wasps. One aphid had the “bright idea” of acquiring a fungal gene through horizontal gene transfer and adopting a yellow color that protects it very effectively against both of these predators.
More recently, a new species of tardigrade identified in China was found to have acquired a gene from a species of bacteria that enables it to protect itself against lethal doses of X-rays. In both of these cases, the organism that provided this genetic gift has been identified because it is still alive; however, this is not possible for the unique genes found in tardigrades.
It appears that tardigrades, which have inhabited our planet for about 600 million years, have had ample time to acquire numerous genes through horizontal gene transfer from species that are now extinct, thereby building up a veritable gene library. This is all the more plausible given that tardigrades have survived the five major mass extinctions of living species that our planet has experienced throughout its history. The most recent of these wiped out the dinosaurs. A small number of these unique tardigrade genes have already been identified and given bizarre names such as Dsup, TDR1, CAHS, SAHS, MAHS, TDPs, LEA, Doda1, and Trid1.
When introduced into human cells or other laboratory organisms (fruit flies, bacteria, yeast, plants, etc.), these genes were able to dramatically increase their resistance to normally lethal treatments such as X-rays, ultraviolet radiation, or powerful oxidants. Better yet, proteins derived from some of these genes have been able to protect drugs from dehydration, allowing them to be stored at room temperature—revealing enormous potential for vaccine distribution without the need for expensive freezers. The future use of these unique tardigrade genes in the biomedical field is already the subject of numerous patent applications, heralding revolutionary new biomedical technologies that could emerge soon—ranging from protecting astronauts’ skin against cosmic rays to the possibility of preserving, through dehydration, drugs, tissues, or organs awaiting use.
A "DNA scent"
But where does this DNA come from that tardigrades can incorporate? The answer lies all around us. We are constantly surrounded by a “DNA signature” released by all the living organisms around us. This DNA is called eDNA, short for environmental DNA. A soil sample, after sequencing the DNA it contains, can reveal which living species inhabit a given location—even without having seen them. This is a highly effective technique for assessing the biodiversity of a terrestrial or marine environment. Recently, scientists successfully identified the DNA signatures of Asian elephants and giraffes from samples taken from a spider web nearly 195 meters away at the Perth Zoo in Australia.
Scientists have proposed a possible scenario to explain how these DNA fragments might end up in tardigrades as well as in certain worms and a few other invertebrates. These organisms all share the ability to survive periods of dehydration of varying lengths.
When they are in cryptobiosis due to dehydration, the gradual formation of breaks in their chromosomes can be observed.
Tardigrades will be able to repair this damage as soon as they are rehydrated. Water is potentially capable of transporting DNA fragments to the cell nucleus, where the chromosomes are located. Their presence among the fragmented chromosomes of tardigrades suggests that these fragments may be integrated as repair mechanisms are at work.
With their ability to acquire new genes from their environment, tardigrades have accumulated genes with exceptional properties from species that have long since disappeared from our planet. These unique tardigrade genes may hold the secrets to future biomedical breakthroughs by offering new possibilities for the protection and transport of fragile drugs and tissues, new safeguards for future missions already planned by space agencies, or even applications in dermocosmetics to combat the effects of aging.
Simon Galas, Professor of Genetics and Molecular Biology of Aging, IBMM CNRS UMR 5247 – School of Pharmacy, University of Montpellier and Myriam Richaud, PhD in Genetics and Molecular Biology of Aging, Faculty of Pharmacy, University of Montpellier
This article is republished from The Conversation under a Creative Commons license. Readthe original article.