Resurgence of the COVID-19 pandemic: Why the Omicron BA.4 and BA.5 variants are spreading in France
After being largely overlooked by the media for several weeks, the COVID-19 pandemic is back in the news as the BA.4 and BA.5 variants take hold in France, following their emergence in Portugal and elsewhere.
Samuel Alizon, French National Research Institute for Sustainable Development (IRD) and Mircea T. Sofonea, University of Montpellier

Mircea Sofonea, an associate professor, and Samuel Alizon, a research director—both specialists in epidemiology and the evolution of infectious diseases within the “Infectious Diseases and Vectors: Ecology, Genetics, Evolution, and Control” unit (University of Montpellier, CNRS, IRD)—analyze the situation in France. What can be said about these variants? Will they lead to a new wave this summer?
The Conversation: The Omicron variant, which has become the dominant strain worldwide, continues to spread and evolve. But its new subvariants are now designated as BA.1, BA.2, then BA.4 and BA.5… How can we keep track of them?
Samuel Alizon: Indeed, it’s easy to get lost in this proliferation of naming systems! The Greek letters were introduced by the World Health Organization (WHO) in 2021 with the Alpha variant. This is probably the worst classification system, as it was developed without taking evolutionary biology into account. The systems used by Pango or Nextclade are much more appropriate. In fact, the WHO seems to have stopped updating its classifications and now groups all BA-type variants under the generic term Omicron.
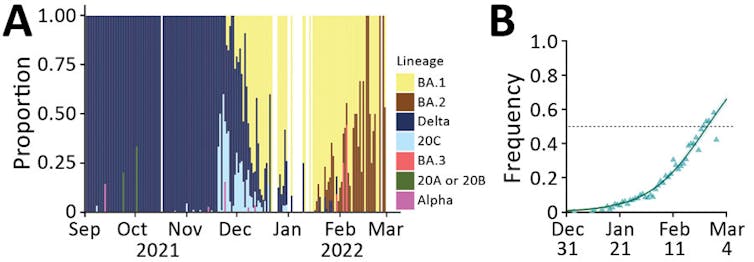
Sofonea et al. (2022, Emerging Infectious Diseases)
In a recent study (see above), we modeled the circulation of variant lineages in France, and the first Omicron wave caused by the BA.1 lineage emerged as early as late 2021. This was quickly supplanted by the BA.2 lineage, which caused a second wave of hospitalizations in April 2022. Now, the BA.4 and BA.5 lineages are taking the lead.
Mircea T. Sofonea: These lineages were identified in May, but they likely emerged in December 2021 in South Africa, possibly from BA.2, which has been the dominant lineage in France since March 2022.
While the BA.2 variant differed from BA.1 as much as the Delta variant differed from the Alpha variant, the evolutionary divergence between BA.4 and BA.5 is more limited.
However, even though the number of new mutations is limited, some are cause for concern. For example, the 452R mutation in the spike protein is known to confer greater affinity for the human ACE2 receptor, which the virus uses to enter our cells. The 486V mutation, also in the spike protein, gives the virus a fairly high capacity for immune evasion.
Nevertheless, caution is warranted when applying analogical reasoning to isolated mutations. This is because the effect of such mutations is neither absolute nor cumulative; it depends on the entire genotype, with potential synergistic and antagonistic effects, including at distant loci on the genome (a phenomenon known as epistasis).
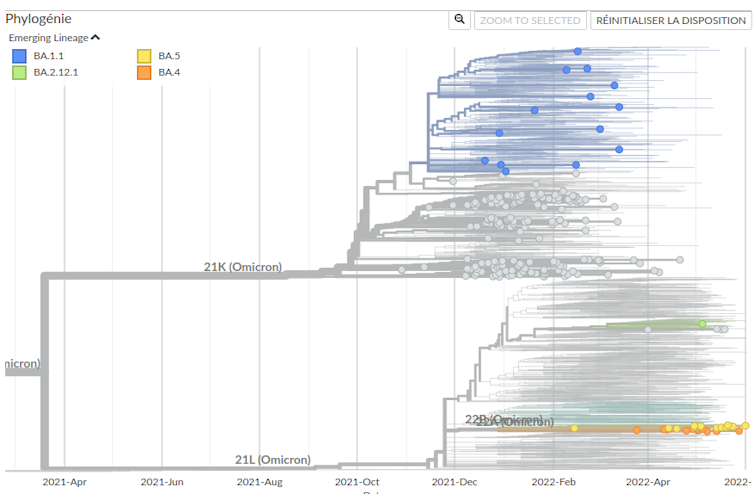
TC: Are these mutations unique to these variants, or do BA.4 and BA.5 “draw” from all the possibilities that have been tested by their predecessors—Delta, Gamma, Beta, and Alpha?
MTS: It’s important to remember that Omicron is not a direct descendant of previous variants, but rather a distant cousin, and that viruses do not mutate in a deliberate or targeted manner. The mutations detected in the genome of a new lineage arose by chance.
The 452R mutation was not present in the BA.1 or BA.2 lineages, but it was found in the Delta variant. In fact, it is one of the three mutations being tested for in the screening tests currently conducted on all positive PCR tests in France.
The 486V mutation is not associated with any of the lineages circulating within our species, but deep mutational scanning experiments—which involve generating proteins with mutations—had identified it as potentially involved in immune evasion.
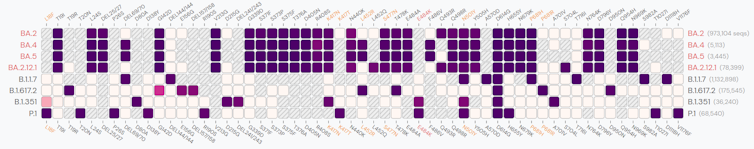
SA: Regarding the differences between variants, two genetic mechanisms are involved: mutations and recombination. The latter allows for the mixing of entire portions of the genome when two viruses from different lineages “co-infect” the same host.
From a biological perspective, several hypotheses exist to explain the emergence of variants: increased circulation within a population, the involvement of an animal reservoir, or chronic infections in immunocompromised individuals. Indeed, these individuals are unable to clear the virus, which consequently leads to longer-lasting and more lethal infections. A preprint (and thus to be taken with caution, as it has not yet been peer-reviewed) by a team in New York describes the intra-patient evolution of a BA.1 virus with the accumulation of key mutations and, most importantly, its transmission to at least five other people.
In the case of BA.4 or BA.5, since their differences from BA.2 are fairly limited, these may simply be mutations that have become established as the virus has circulated.
TC: Why are BA.4 and BA.5 spreading in France right now?
SA: It is easy to estimate the growth advantage of one lineage over another within a population. According to our team, BA.5 has a growth advantage of about 9% over BA.2 in France.
However, it is difficult to determine the source of this advantage. Is BA.5 spreading more widely because it is more contagious? Or is it because it evades immunity more effectively? A preprint by a Japanese team and a publication by a Chinese team highlight the role of immune evasion, particularly through the 486V mutation.
Regardless of the source of this advantage, it could contribute to a resurgence of the epidemic in France.
MTS: A second mechanism is also at work in France: immunity against SARS-CoV-2—which is essentially hybrid, meaning it results from both vaccination and infection—declines over time following the most recent immunogenic event (whether an infection or vaccination).
While the protection provided by an Omicron infection or athird vaccine dose remains significant after five months against severe disease, it is, however, greatly reduced against infection in general. The population’s susceptibility to the virus (i.e., the counterpart to herd immunity) therefore recovers over time, eventually opening the door to a resurgence of the epidemic.
In short, BA.4 and BA.5 are spreading as our immunity wanes, and they are doing so more rapidly than BA.2 because they have the dual advantage of higher transmissibility and greater immune evasion. BA.4 and BA.5 are therefore triggering a wave sooner than BA.2 would have.
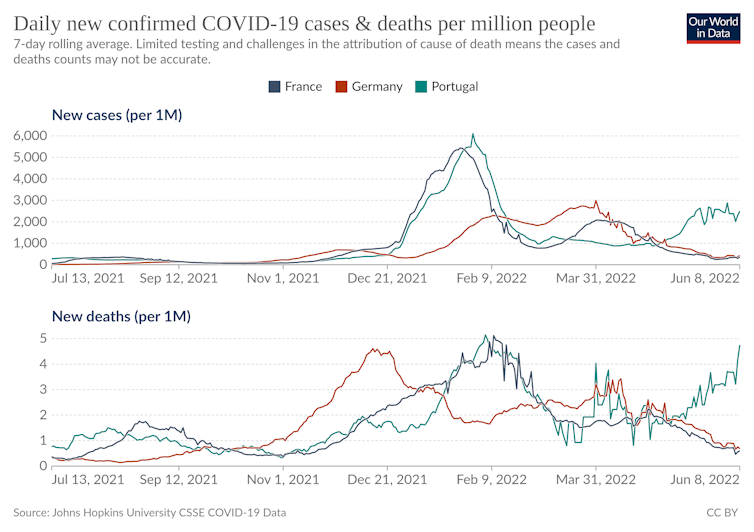
TC: The situation in Portugal may have been cause for concern. But can we learn anything from the trends observed in other countries?
MTS: I am cautious about cross-country comparisons: they are becoming increasingly difficult, as current transmission rates depend not only on the health measures in place but also on epidemiological and immunological histories, which vary increasingly from country to country.
In France, it remains difficult to quantitatively compare the easing of restrictions—which is contributing to the economic recovery—with the summer season, which is limiting that recovery, as longer, warmer days encourage social interactions in open-air settings.
SA: Portugal is one of the European countries where the BA.4/BA.5 wave is most advanced and is accompanied by a rise in hospitalizations. It is difficult to say why it began there so early, but, as with all the onset of epidemics, random events such as “super-spreader” events likely play a significant role.
Globally, in South Africa, the BA.4/BA.5 wave appears to be on the decline. In the United States, however, BA.2 was initially replaced by the BA.2.12 lineage, but this lineage now appears to be on the verge of being replaced by BA.5.
TC: Can we predict how these shifts between variants will affect future epidemic peaks?
SA: In 2021, in France, a new variant replaced the older ones because it was more contagious. Since December 2021, it has been immune escape that has taken center stage.
This makes modeling scenarios challenging. The models developed by our team, as well as those fromthe Pasteur Institute and the Pierre Louis Institute of Epidemiology and Public Health, already took into account vaccination coverage in the population and the percentage of people who had been naturally infected.
On the other hand, factoring in the time that has elapsed since the last vaccination or natural infection is a challenge, because after two years of the pandemic, two vaccination campaigns, and a massive BA.1 wave, everyone now has a different level of immunity!
MTS: We have developed tools to account for this heterogeneity in immunity across populations. Given our constraints, we are currently focusing on the long term, but, in theory, it should be possible to use this framework to explore short-term scenarios.
At this point, it is difficult to say exactly how severe the new wave of the epidemic that is beginning will be. This wave, in the genetic or virological sense, is already well underway, and BA.5 will likely become the dominant variant by June 20. While we can count on summer to reduce the incidence compared to winter, that alone will not prevent a wave of infections. For the record, one of the peaks of circulation in France remains August 2020, and the 4the The (Delta) wave had peaked in July 2021.![]()
Samuel Alizon, Director of Research CNRS, French National Research Institute for Sustainable Development (IRD) and Mircea T. Sofonea, Associate Professor of Epidemiology and the Evolution of Infectious Diseases, MIVEGEC Laboratory, University of Montpellier
This article is republished from The Conversation under a Creative Commons license. Readthe original article.