How the Omicron BA.2 variant has pushed the initial boundaries of COVID-19
Although the COVID-19 pandemic has taken a back seat in the news, it is by no means over. SARS-CoV-2 continues to circulate and evolve rapidly. Researchers at the “Infectious Diseases and Vectors: Ecology, Genetics, Evolution, and Control” unit (University of Montpellier, CNRS, IRD), Mircea Sofonea, associate professor, and Samuel Alizon, research director—both specialists in the epidemiology and evolution of infectious diseases—take stock of Omicron now that it has spread across the globe. How can we explain its subvariants, and what do we now know about its capabilities and the effectiveness of our immune system against future variants?
Samuel Alizon, French National Research Institute for Sustainable Development (IRD) and Mircea T. Sofonea, University of Montpellier
The Conversation: What is the current situation regarding the spread of SARS-CoV-2 variants in France and more broadly?
Samuel Alizon: Since December 2021, the Omicron variant—specifically the BA.1 lineage (or 21K, in the Nextstrain classification)—has replaced the Delta variant (B.1.640 lineage) worldwide. This replacement occurred even more rapidly than the shift from the Alpha variant to Delta during 2021. In France and elsewhere, its BA.1.1 sublineage has also been spreading since January.
Even more unexpectedly, in some countries—including Denmark, South Africa, and India—a related lineage known as BA.2 has rapidly become the dominant strain. While the divergence of BA.1.1 from BA.1 is recent (around October 2021), the common ancestor of BA.1 and BA.2 is believed to date back to March 2021. In summary, these two lineages share a common origin (they share more than 30 mutations) but are already nearly as divergent as Delta was from Alpha (by more than 35 mutations).
In France, thanks to a collaboration with the CERBA laboratory and Montpellier University Hospital, our team estimated that the Omicron/BA.1 variant had become the dominant strain during thethird week of December and had become the predominant strain shortly thereafter. Since late January, the percentage of BA.2 cases among test results has doubled every ten days in January, becoming the dominant strain by the end of February.
newsnodes.com/omicron_tracker/Questzest, CC BY-SA
T.C.: Beyond BA.1 and BA.2, there’s talk of a third subvariant within the Omicron group: BA.3. What’s the story there?
Mircea T. Sofonea: The Omicron BA.3 sublineage has been known since November 18, 2021, when it was first sequenced in South Africa—less than a month after BA.1 was detected in Botswana. It has 34 mutations in its spike protein (which is also shortened by six amino acids) compared to the initial SARS-CoV-2 reference strain (Wuhan-Hu-1), a number intermediate between the BA.1 (39) and BA.2 (31) sublineages, relative to which it has only a single unique mutation.
Based on these arguments, some authors suggest that BA.3 originated from a recombination between BA.1 and BA.2.
Although it is present in at least twenty countries (in southern Africa, Europe, and the United States) and, based on in silico analyses, appears to have a mutational profile that makes it more transmissible than BA.1, it remains extremely rare. In fact, only 543 BA.3 sequences had been submitted to the GISAID database as of March 5, 2022, representing less than 0.05% of global Omicron sequences. In France, 17 BA.3 sequences had been reported as of February 28, 2022, most of which belonged to the same cluster.
This low transmission rate makes it impossible at this stage to accurately quantify its transmission advantage or any difference in virulence, but it does offer a reason for optimism regarding the risk this sublineage poses in the short term. This rules out its classification as a variant in its own right.
T.C.: The fact remains that this “Omicron group” exhibits significant diversity… More so than what was observed in previous variants? Can we conclude that there are, in fact, several Omicron variants?
M.T.S.: For now, the World Health Organization’s (WHO) designation “Omicron variant” does indeed refer to all the lineages BA.1, BA.1.1, BA.2, and BA.3… But it is indeed important to highlight the differences not only between the Omicron group and other variants, but also between BA.1 and BA.2, which are significant from both virological and epidemiological perspectives.
It is important to note—contrary to the popular misconception of linear viral evolution—that the Omicron group did not originate from the Delta variant but likely emerged around the same time: that is, in the early months of 2020, either throughchronic infection of an immunocompromised host or via a zoonotic transmission from a murine reservoir.
These two hypotheses would indeed explain the significant accumulation of mutations in this group at the time of its detection: at least 31 in the spike protein alone, more than double that of the Delta variant.
Structural and functional studies of the Omicron spike protein (using high-resolution cryo-electron microscopes, which France sorely lacks) have revealed the numerous (synergistic) effects of these mutations on its virology, pathogenicity, and epidemiology.
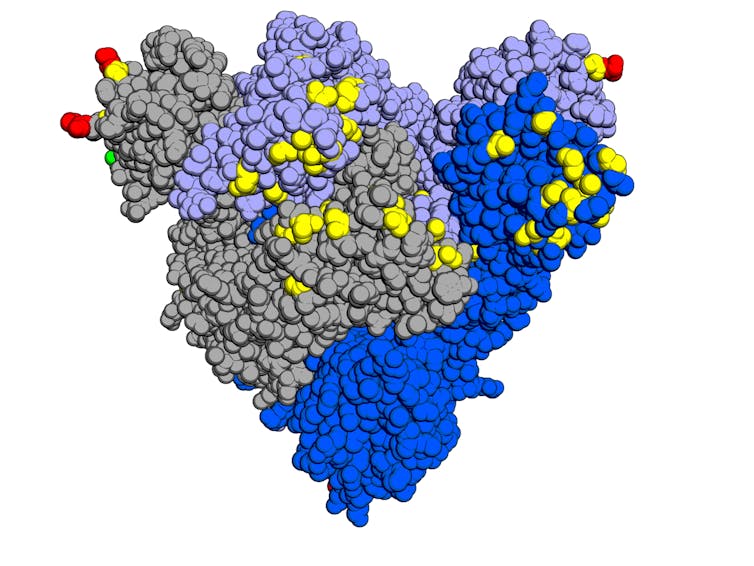
Opabinia regalis, based on “Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein,” in Cell, CC BY-SA
T.C.: Now that we have a little more perspective, what insights can we gain from studying these changes?
M.T.S.: Indeed, many of Omicron’s capabilities have been identified or clarified:
● Increased affinity for the human ACE2 cell receptor but reduced membrane fusion capacity,
● Utilization of a second pathway for entry into cells via endocytosis (independent of an enzyme required forthe entry of other variants),
● Increased replication potential in the epithelium (tissue composed of tightly packed cells) of the upper airways (mouth, nose, throat, etc.) and decreased replication potential in that of the lower airways (lungs, etc.),
● The inter-infection interval (serial interval) has been shortened from 4.1 (Delta) to 3.7 days,
● Immune escape, leading to a significant reduction in neutralization by antibodies derived from infection, vaccination, or therapeutic treatment (monoclonal antibodies).
The combination of these characteristics gives Omicron two to three times the transmissibility and a fatality rate that is approximately 70% lower than that of the Delta variant.
It should be noted, however, that this decrease in virulence is not consistent across all age groups: British researchers have shown that the risk of hospitalization due to Omicron infection is equivalent to that of Delta among children under 10, but four times lower among those aged 60–69.
T.C.: What about vaccination against Omicron?
M.T.S.: According to the latest UK public briefing, vaccine efficacy against symptomatic infection with Omicron is low and declining rapidly. With only two doses, protection drops to just 10% after 6 months, compared to over 40% for Delta. However, it remains above 40% for up to 6 months after thethird dose. Fortunately, vaccine efficacy against hospitalization exceeds 75% for up to six months after the booster shot.
Based on current knowledge, these estimates are equivalent for the two main Omicron sublineages, BA.1 and BA.2, which exhibit comparable disease severity. The two notable differences between them lie in a 40% relative transmission advantage for BA.2 —linked to faster replication in the nasopharynx and an even shorter serial interval (3.3 days)—as well as a lack of detectable neutralization by the only monoclonal antibodies still active against BA.1, namely sotrovimab and the tixagevimab/cilgavimab combination therapy.
T.C.: Are we still well protected against new variants?
M.T.S.: Between post-infection immunity and vaccine-induced immunity, less than one-tenth of the French population is now immunologically “naive” to the COVID-19 virus.
The Omicron BA.1 wave has conferred cross-protective herd immunity against BA.2, with reinfections being rare. However, this immunity is only partially temporary, as we see with the vaccine.
Since SARS-CoV-2 cannot be eradicated (due to its high transmissibility, imperfect and waning immunity, and animal reservoirs), new variants will emerge, selected in particular for their ability to evade the immune response. The timing of their emergence and their antigenic properties are currently unpredictable.
Booster shots—ideally using updated or more effective vaccines—will remain necessary, at least for vulnerable individuals. In fact, we should already be working on optimizing the schedule for future vaccination campaigns and establishing criteria for the temporary, localized reintroduction of preventive measures in the event of a resurgence of the epidemic.
T.C.: You mention that new variants will continue to emerge. Is it possible for different variants to coexist?
S.A.: That will depend largely on cross-immunity. According to population dynamics theory, in the simplest scenarios, two species cannot coexist if they occupy the same “ecological niche.” The Omicron variants appear to mark a turning point by colonizing a new “niche” in the upper respiratory tract.
From an immunological perspective, this coexistence could be facilitated by the fact that the immunity generated following infection with the Delta variant or vaccination extends only minimally to the upper respiratory tract, which is poorly supplied by the immune system. Furthermore, preliminary findings suggest that natural immunity following infection with the Omicron variant offers little protection against other variants.
Finally, we need to clarify the context in which we are discussing coexistence. We know that two variants can coexist within the same person, as demonstrated by the existence of a recombinant virus involving the Alpha variant and other lineages, or by recent cases of co-infection with Omicron and Delta in the United Kingdom.
Conversely, since the start of the pandemic, we have seen certain variants remain dominant in specific countries only. Here again, ecological science teaches us that geographical constraints facilitate the coexistence of species.
T.C.: Does the emergence of Omicron and BA.2—which are outpacing all other variants—signal that the epidemic is “running out of steam” in terms of its ability to mutate?
S.A.: From the perspective of viral evolution, it’s actually the opposite. It’s striking to see that BA.2 differs from the SARS-CoV-2 sequence from Wuhan by nearly 80 mutations, whereas BA.1 has 70 and Delta “only” 50.
The more a virus spreads, the faster it evolves. And the Omicron variant is spreading extremely quickly… Another example of this widespread circulation comes from the limited monitoring of animal reservoirs: analysis of sequencing data suggests that cryptic lineages appear to be infecting the wildlife in New York City’s sewers.
The increasing “biodiversity” of this virus undermines hopes of reaching “evolutionary dead ends”—that is, situations where the mutations necessary for its spread (such as those enabling it to evade the immune system) are so costly that they cannot propagate. Indeed, there is a high risk that one of the countless lineages will find a viable solution.
We should also be wary of the long-held belief that viruses become less virulent over time. As demonstrated by this year’s discovery of an HIV variant—more virulent than average—that has been circulating since the late 1990s, the opposite is actually true. While the fatality rate of infections is declining, this primarily reflects improvements in public health responses, vaccines, and treatments.
One lesson from modeling is that we must be wary of our perceptual biases. During the growth phase of an epidemic, anxiety rises, and conversely, during the decline phase, we tend to be more optimistic… The challenge for scientists is to stick to the facts. From this perspective, we still lack a great deal of certainty regarding the Omicron variants. What is the duration and scope of post-infection immunity? What are the long-term effects of infection? Will we see, as with other variants, cases of long COVID emerge in the coming months?
On all these key issues, we are eagerly awaiting the findings of research teams abroad, which have far greater resources at their disposal.![]()
Samuel Alizon, Director of Research CNRS, French National Research Institute for Sustainable Development (IRD) and Mircea T. Sofonea, Associate Professor of Epidemiology and the Evolution of Infectious Diseases, MIVEGEC Laboratory, University of Montpellier
This article is republished from The Conversation under a Creative Commons license. Readthe original article.