COVID-19: A New Modeling Method to Better Assess Epidemic Risks
For thousands of years, humanity has been struck by epidemics… Each time, people have taken protective measures that reflected the knowledge—and even the beliefs—of the era regarding modes of transmission. Take, for example, the plagues that ravaged Europe a few centuries ago. At the time, it was firmly believed that this devastating disease was caused by inhaling miasmas and the resulting imbalance of bodily humors. This led to the widespread use of masks stuffed with medicinal herbs and fitted with a pointed beak: this is the classic image of bird-beak masks…
Simon Mendez, University of Montpellier and Alexandre Nicolas, Claude Bernard University of Lyon 1

At the beginning ofthe 20th century, there was a major shift toward recognizing the predominant role of close contact in transmission. Recommendations changed, and the preventive measures promoted at the start of the COVID-19 pandemic stemmed from this shift: handwashing, possibly with hand sanitizer, using disposable tissues, sneezing into one’s elbow, and so on. All of this is intended to mitigate the risk of direct transmission via the hands or through objects contaminated (known as fomites) by an infectious person.
Over the past three years of the pandemic, the academic understanding of how respiratory diseases spread has undergone a major shift. What do we currently know about the mechanisms involved? How can we assess risks based on different situations—such as a café terrace, a line where social distancing is observed, or a busy street? We sought to answer these questions by developing a simple and rapid modeling method.
Shedding light on the mechanism of aerosol transmission
At this time, there is reason to believe that aerosol transmission is the primary mechanism by which COVID-19 spreads.
On this research track, the collection of experimental data had begun well before the pandemic: images from Lydia Bourouiba’s group on the projection of microdroplets emitted during a sneeze (below), as well as those from the teams led by Lidia Morawska and William D. Ristenpart, among others, on the size of droplets and aerosols emitted during various exhalation activities, date back several years.
These studies show that coughing and sneezing can project aerosols over distances potentially exceeding 2 meters, and that simply speaking for one minute can generate as many droplets as a bout of coughing.
Despite these findings, disagreements persist regarding basic data such as the size distribution of the droplets and aerosols produced—a factor that is nonetheless critical for determining their fall time and likelihood of remaining suspended in the air indoors. Furthermore, experimental difficulties and ethical concerns prevent the study of all possible emission, inhalation, and environmental conditions.
Computer simulation: the solution?
So how can we increase the number of possible tests? One solution is to use digital tools, leveraging the computational power of computers.
The next step is to choose which system to simulate: airborne transmission occurs through the transport of viruses within droplets and aerosols formed in the respiratory tract (lined with mucus) and the mouth (filled with saliva), the challenge is to simulate the spread of these virus-carrying microdroplets from their emission through to their inhalation—and even their penetration and deposition in the respiratory tract.
Such fluid dynamics simulations have become increasingly popular since the onset of the pandemic and have highlighted the complexity of the process, the importance of accurately describing the turbulent structures of the flow, the variability of the exhaled airflow depending on phonation, the sensitivity of droplet evaporation to the breath environment, and so on.
If we rely solely on crude models, the assessment of risks can be significantly compromised, and in the early stages of the epidemic, we saw numerous studies based on questionable assumptions. Conversely, highly detailed models, which sophisticatedly simulate the spread of respiratory droplets, offer greater realism… But they run into difficulties with the complexity of analyzing the data produced (what to do with these ranges of trajectories that vary with every micro-change?) and with their computational cost—the crux of the matter for numerical simulations.
The "dynamic risk maps" solution
To get the best of both worlds, one approach is to use highly detailed fluid dynamics simulations—with a resolution on the order of millimeters—and to estimate the risk dynamics around an emitter at a more aggregated level, that is, without concerning oneself with the precise location of each individual droplet.
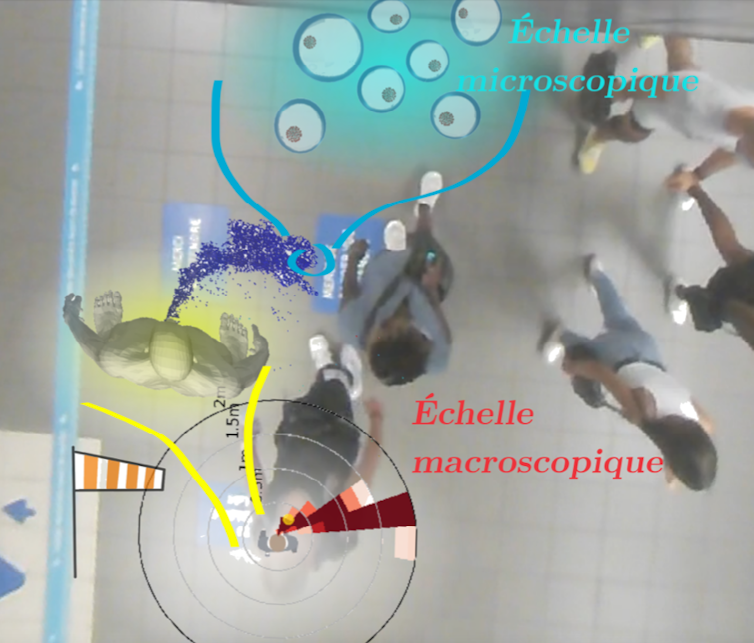
Except that a single map isn’t enough: in reality, the resulting “risk map” varies depending on whether the person is speaking or walking, or based on wind or drafts, and so on. We therefore need to build a whole library of reference scenarios and, in the gaps between them, infer the missing ones. That is what we have set out to do.
The results provide clear and straightforward insights. It turns out that even the slightest breeze in the area drastically reduces the risk of viral transmission. This puts an end to a debate that began at the start of the pandemic, when people wondered whether wind might actually promote transmission by carrying droplets and aerosols further. In fact, in all the scenarios studied, it disperses them.
More generally, the significant reduction in computational costs made possible by the use of “dynamic risk maps” has made it feasible to analyze real-world scenarios involving dozens or even hundreds of people.
Tangible results
In practical terms, we were able to walk the streets of Lyon in the midst of the pandemic and set up our camera equipment (filming people from above while respecting their anonymity) in various locations, both outdoors and in indoor spaces that were not overly confined (spacious and well-ventilated). These included an SNCF train station, a metro station, busy streets, an open-air market, café terraces, and a developed riverside area along the Rhône.

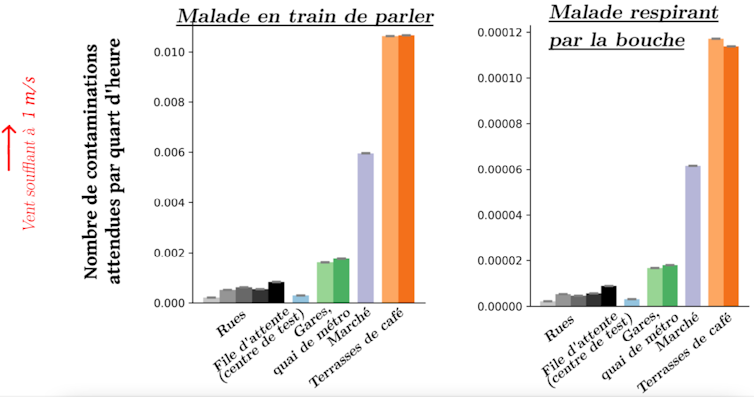
From these videos, we extracted the trajectories and orientations of pedestrians’ heads and overlaid this data onto the aforementioned risk maps (Figure 1). Results:
- Busy (but not crowded) streets pose a very low risk compared to the open-air market, where there were far more people and they were much closer together. As might be expected, density plays a major role.
- All of these situations posed a lower risk of new infections than café terraces (Figure 3), where people have close and prolonged contact, even though the overall density is lower there.
- Exhalation plays a major role, since the amount of droplets emitted by a person speaking is much higher than when they are breathing through their mouth (and, even more so, through their nose).
This prioritization of real-world scenarios based on theoretical and numerical models illustrates how high-fidelity simulations can be used to examine everyday situations. This can serve as a decision-making tool in public health policy.
The modeling tool thus serves as a valuable bridge between fundamental knowledge about airborne viral transmission and the public health measures that need to be implemented.
Strengths and limitations of a promising approach
Compared to detailed approaches, in which the trajectory of each respiratory droplet is predicted and influenced by numerous factors (air recirculation around street furniture, the wake effect of each pedestrian, etc.), our method allows us to estimate risks in real-world situations in just a few minutes, since the most computationally intensive simulations have already been carried out once and for all.
Admittedly, this comes at a cost: the impact of pedestrians other than the transmitter is not taken into account, which proves to be a limitation in extremely dense crowds. Other factors, such as the effect of temperature on the dispersion of exhaled aerosols, could, however, be incorporated into future improvements.
Despite these limitations, the model has a wide range of applications, as it can be combined with field measurements for risk prioritization or integrated with simulated pedestrian trajectories. For example, it can be used to quantify in advance the impact of architectural choices on transmission risks within a building or to manage crowd flow during an epidemic.
Simon Mendez, Research Fellow at the CNRS, Mathematics and Modeling Laboratory, University of Montpellier and Alexandre Nicolas, Research Fellow at the CNRS; physicist, Claude Bernard University Lyon 1
This article is republished from The Conversation under a Creative Commons license. Readthe original article.