Why it is unlikely that the SARS-CoV-2 coronavirus will lose its virulence
Since the start of the COVID-19 pandemic, strong opinions have regularly been expressed regarding changes in the virulence of SARS-CoV-2. Many believe that its virulence must inevitably decrease, since viruses, bacteria, and other pathogens have historically lost their virulence by adapting to their hosts.
Samuel Alizon, French National Research Institute for Sustainable Development (IRD) and Mircea T. Sofonea, University of Montpellier

Unfortunately, this “intuition” does not hold up to scrutiny, as it requires us to view the human immunodeficiency virus (HIV), the tuberculosis bacillus, the malaria parasite, and the influenza virus as exceptions. In fact, no matter how hard one looks, it is difficult to find “parasites” (to use a term from evolutionary biology) that conform to this axiom, dubbed the “law of decreasing virulence” in the early 20th century.
Why, then, does this view persist? What do recent discoveries in evolutionary biology tell us about virulence? And what can we expect in the case of SARS-CoV-2?
Do not confuse lethality with virulence
The logic behind the theory that parasites systematically evolve toward anon-virulent state is child’s play: for the parasite, killing its host is like killing the goose that lays the golden eggs. In other words, strains (or “variants,” to use a more fashionable term) that kill their host quickly should be less successful than others, and thus disappear.
One reason this century-old theory remains so influential is the confusion between the concepts of lethality and virulence.
Lethality is the proportion of infected individuals who die as a result of infection by a given parasite, in a given location, at a given time. Many factors contribute to reducing apparent lethality: treatments, vaccination, quality of clinical care, etc. Virulence, on the other hand, refers to the parasite’s propensity to harm its host. It is quantified in the absence of specific treatment.
In other words, the same viral variant will have a different fatality rate from one country to another, depending, for example, on the quality of the healthcare system. However, its virulence will remain unchanged.
In the case of SARS-CoV-2, we have seen a decline in case fatality rates since the start of the pandemic in many countries, largely thanks to vaccination. In contrast, virulence has increased. Infections with the Alpha variant are more likely to result in death than those involving the ancestral lineages that were circulating in early 2020. As for the Delta variant, early findings suggest it is more virulent than the Alpha variant, as it appears to lead to more hospitalizations among unvaccinated individuals. Preliminary results point in the same direction for the Beta variant.
Let’s admit that this is counterintuitive. But it also illustrates that evolutionary biology is a discipline in its own right, and that it is risky to proclaim oneself an expert in it. Yet many view it instead as a sort of “hobby,” something one might pursue after gaining sufficient experience in more fundamental subjects, such as the mechanisms of development or the physiology of organisms. This is likely another reason for the persistence of misconceptions regarding the evolution of virulence.
Does evolutionary biology really have more to offer than the insights of “wise elders”? Obviously, this field of research suffers from the fact that we cannot replicate an epidemic exactly. However, the analysis of epidemics that preceded the one we are currently experiencing (notably the HIV epidemic) and so-called “experimental” evolutionary studies conducted on other parasites offer valuable insights.
Two types of virulence
To begin with, we must distinguish between two categories of damage inflicted by a parasite on its host, depending on whether or not they influence the parasite’s spread: non-adaptive virulence and adaptive virulence.
Non-adaptive virulence benefits neither party. In the case of SARS-CoV-2, this includes particularly severe manifestations of the infection, such as cytokine storms. The second category of virulence is referred to as “adaptive” because it is associated with increased transmission of the pathogen, either directly or indirectly.
Seth Pincus, Elizabeth Fischer, and Austin Athman, National Institute of Allergy and Infectious Diseases / NIH
In the case of HIV, for example, it has been shown that more virulent variants—those that produce more viral particles by exploiting their host cells more efficiently or evading the immune response more effectively—are also more contagious, since the likelihood of transmission is linked to the amount of virus in the blood.
Distinguishing between non-adaptive and adaptive components of virulence allows for a better understanding of the evolution of this trait. It is generally expected that non-adaptive virulence will decrease, since it is associated only with costs. However, it is not that simple, as the parasite’s life cycle must be taken into account.
In the case of SARS-CoV-2, severe symptoms generally appear two weeks after infection, yet more than 95% of transmissions occur before the11th day. In other words, from the perspective of this coronavirus, late pathological manifestations (particularly inflammatory ones) of virulence do not result in a loss of transmission opportunities. Consequently, it is unfortunately unlikely that natural selection will favor variants that cause such immunopathological manifestations less frequently.
When it comes to the adaptive component of virulence, the picture is even less straightforward. Everything depends on the relationship between the costs (virulence) and the benefits for the virus’s spread (transmission rate, duration of infection). In the case of HIV, again, it has been shown that an intermediate level of virulence maximizes the virus’s selective value—that is, the number of infections caused by a person carrying the virus.
One factor that might lead us to believe there is such a correlation between virulence and transmissibility in the case of SARS-CoV-2 is that more transmissible variants are also more virulent.
For example, our team has shown that in France, the Alpha variant had a clear advantage in terms of transmissibility compared to ancestral lineages. Our British colleagues, for their part, concluded that its virulence had increased by 50%. Similarly, in June 2021, we demonstrated that the Delta variant was more contagious than the Alpha variant. Following this logic, a more transmissible variant could therefore be even more virulent.
What can we expect?
The fact that more contagious variants are also more virulent suggests that the adaptive component of virulence is not negligible.
The challenge in predicting how the virus will evolve is that we need to know to what extent the virus—specifically the Delta variant—has now adapted to us. Does this coronavirus still have room to exploit its human host more effectively (in other words, given this level of transmissibility, could it be less virulent)? Or, on the contrary, would any decrease in virulence also result in a decrease in transmissibility?
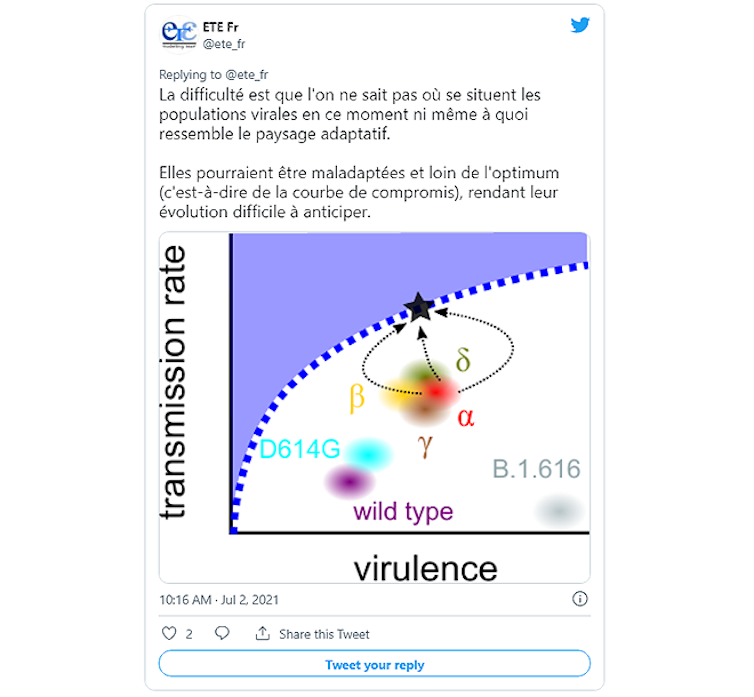
@ete_fr – July 2, 2021
In the first case, if the virus retains some flexibility to better adapt to its host, it is virtually impossible to predict the next stage of its evolution. Mutagenesis experiments could nevertheless help identify which viral forms are the most stable. It should be noted that these generations of mutants in the laboratory are produced in a safe manner (for example, work will be limited to a specific viral protein).
Such studies, known as “deep mutation screening,” have been conducted on a portion of the gene responsible for producing the virus’s S protein (the part containing the genetic information for the receptor-binding domain, or RBD), which acts as a “key” for the virus to enter our cells. This involves generating all possible mutations in the RBD and then studying their effect on the production of the resulting S proteins, as well as their ability to bind to the ACE2 receptor located on the surface of cells (the receptor that serves as the virus’s entry point). This research has identified sites that are particularly at risk in terms of variant evolution.
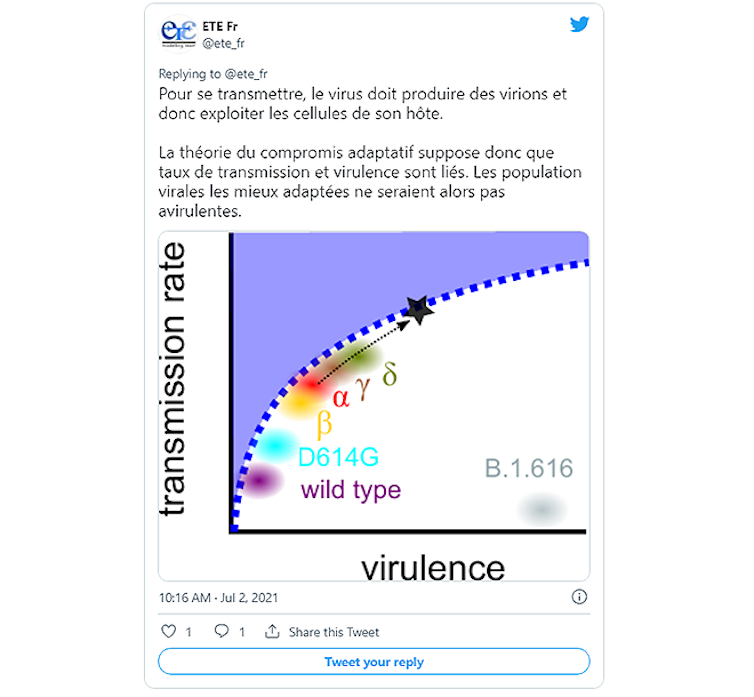
@ete_fr – July 2, 2021
In the second case, one might expect virulence to stabilize at a level that maximizes the number of secondary infections—in other words, an optimization between transmission rate and virulence. Predicting this would require knowing exactly the trade-offs between these two aspects of the infection. But, again, that’s not the end of the story.
The Effects of Immunization
Population immunity (whether natural or acquired through vaccination) radically alters the adaptive landscape of viruses: the most adapted variant in an unprotected population may become a minority in an immunized population. Brazil experienced this in a tragic way, as despite having built herd immunity at the cost of a major health crisis, it still fell victim to a second wave caused by the Gamma variant—which is believed to have rapidly spread throughout the country because it was partially capable of evading the immunity conferred by ancestral strains.
NIAID / NIH
In the short term, vaccination is essential: like natural immunity, it reduces the fatality rate of the infection, accelerating the transition toward a pattern similar to that of seasonal respiratory viruses. Indeed, a year ago, no one would have dared to hope for such vaccine efficacy against severe disease (even for the Delta variant, this protection is estimated to be around 85%). This effectiveness highlights vaccine inequities all the more: for those who will not be fortunate enough to have access to the vaccine, the fatality rate of infections is already higher than in 2020 and could rise further as future variants emerge.
Reconciling Pasteur and Darwin
Beyond the sense of urgency stemming from the health crisis, it is important to consider the pandemic from a medium- and long-term perspective. Making predictions is very difficult, because the relationship between the virus and our immune system is part of a co-evolutionary process: viruses mutate, and our immune responses change. To try to anticipate the future, we must take into account the effectiveness and duration of natural and vaccine-induced immunity, which slow the rate of evolution of viral populations: fewer infections mean fewer mutations…
The study of these dynamic relationships and their implications is the focus of evolutionary biology. Unfortunately, in addition to the chronic lack of funding for scientific research in France, this discipline suffers from a lack of recognition, and the available knowledge is too rarely put to use. One example among many: despite efforts to include various aspects of SARS-CoV-2 research within the scientific advisory board, the board does not include a single evolutionary biologist.
Relying on the insights of “traditional wisdom”—which is unaware of the latest discoveries and tools in evolutionary biology—to predict how pathogens will evolve is like moving forward with our eyes closed. When it comes to infectious diseases, this can have particularly serious consequences given the speed at which microbes evolve and spread. In public health, there is an urgent need to reconcile Louis Pasteur and Charles Darwin.
For more information:
– Alizon, S. & Sofonea, M. T. (2021) SARS-CoV-2 virulence evolution: Avirulence theory, immunity, and trade-offs. Journal of Evolutionary Biology
– Alizon S. (2020) Pandemics, Ecology, and Evolution, Points Publishing![]()
Samuel Alizon, Director of Research CNRS, French National Research Institute for Sustainable Development (IRD) and Mircea T. Sofonea, Associate Professor of Epidemiology and the Evolution of Infectious Diseases, MIVEGEC Laboratory, University of Montpellier
This article is republished from The Conversation under a Creative Commons license. Readthe original article.